
Advancing Orthopedic Care Through Innovation
AK Medical Holdings Limited (HKEX: 01789.HK) is the world's first company to commercialize 3D printing technology in orthopedic joint replacement, spine repair, and trauma fixation implants. Trusted by 7,500+ hospitals across 40+ countries.
World Leader in 3D-Printed Orthopedic Implants
Founded in Beijing in 2003, AK Medical pioneered the global commercialization of 3D printing in orthopedic surgery, transforming how surgeons restore mobility to patients worldwide.
Our Mission
"Make you move for a better life."
We excel to be a world-class business organization through innovation, delivering cutting-edge orthopedic solutions that improve patient outcomes globally.
Our Core Values
Integrity
Customer first, honesty, seeking truth from facts
Innovation
Bravely embracing change and making a difference
Sharing
Willing to share experiences and success
Passion
Pursuing progress with passion every day
Our Journey
Founded in Beijing
AK Medical established as orthopedic implant manufacturer
Hong Kong IPO
Listed on HKEX Main Board (01789.HK)
JRI Acquisition
Acquired JRI Orthopaedics Ltd. in Sheffield, UK
Libeier Integration
Acquired Beijing Libeier for spine & trauma implants
Our Group Companies
AK Medical operates through a network of specialized subsidiaries across China and the UK
Beijing AKEC Medical
Core R&D and ManufacturingNational 'Little Giant' High-Tech Enterprise, operating CNAS-accredited testing center
ITI Medical (Changzhou)
Large-Scale ManufacturingHigh-volume production facility for complete implant and instrument portfolio
JRI Orthopaedics (UK)
Biologic Coating TechnologyUK's largest biologic-fixation implant manufacturer, Queen's Award winner
Beijing Libeier
Spine & Trauma ImplantsPioneer in China's spine implant industry since 1996
Four Pillars of Innovation
Our proprietary technology platforms represent decades of R&D investment and clinical validation, setting the global standard for orthopedic implant innovation.
3D Printing Precision Building Technology
Proprietary additive manufacturing platform producing porous titanium implants with interconnected trabecular structure matched to natural cancellous bone for superior osseointegration.
Clinical Applications:
- Hip cups
- Spinal cages
- Patient-matched tumor implants
Vacuum Plasma Spray (VPS) Coating
Advanced hydroxyapatite (HA) bio-coating technology for enhanced bone-to-implant bonding, developed by JRI Orthopaedics with 50+ years of clinical heritage.
Clinical Applications:
- Femoral stems
- Acetabular components
- Revision implants
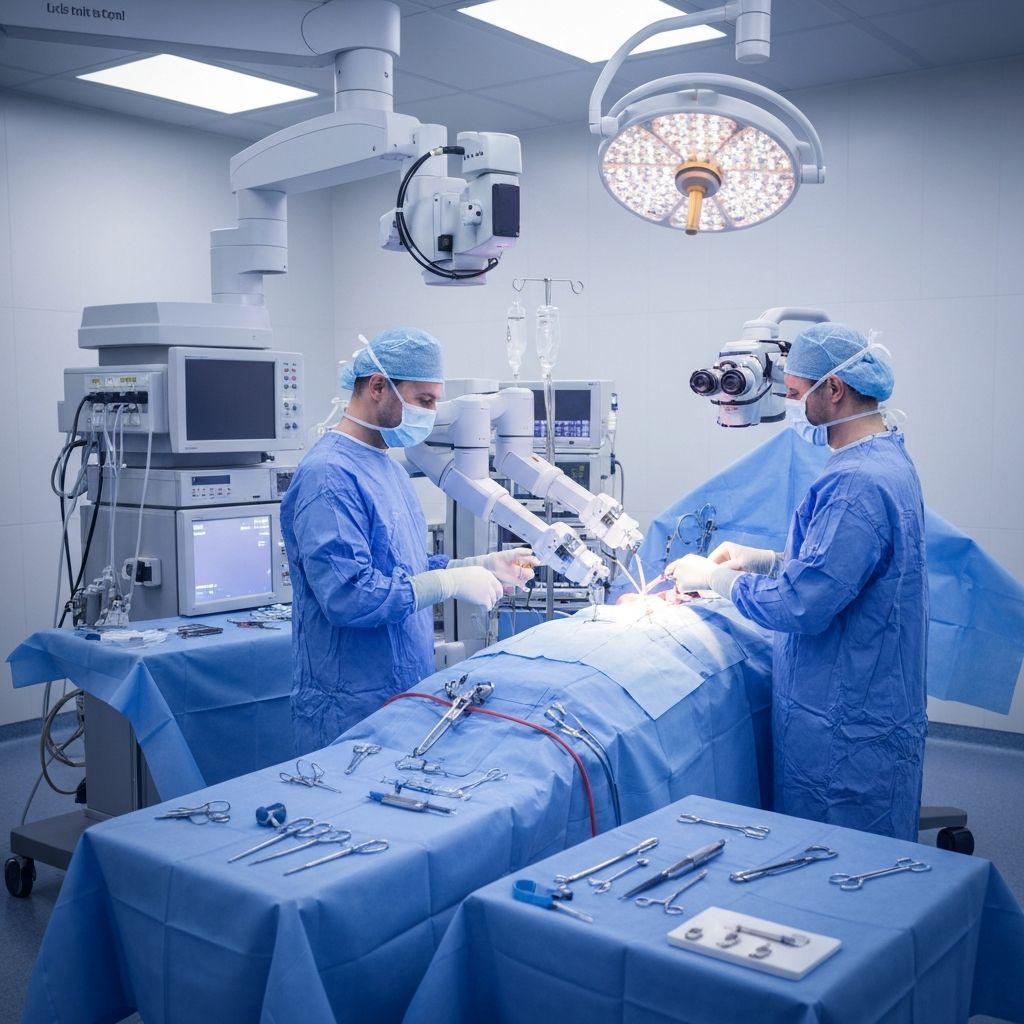
Navigation & Robotic Intelligence
Integrated surgical navigation and robotic assistance platform providing real-time tracking and sub-millimeter implant placement accuracy.
Clinical Applications:
- Robot-assisted THA
- Computer-guided TKA
- Intraoperative navigation
Innovative Custom Orthopedic Solution
End-to-end personalized implant pathway from imaging to surgical delivery, enabling complex reconstructions that standard implants cannot address.
Clinical Applications:
- Tumor reconstruction
- Severe revisions
- Congenital deformities
Comprehensive Orthopedic Solutions
From hip and knee replacement to spine surgery and tumor reconstruction, AK Medical provides the most complete range of 3D-printed orthopedic implants globally.

Hip Replacement
Cementless and cemented total hip arthroplasty systems with 3DACT™ technology
- 3DACT™ porous titanium acetabular cup
- Supravit™ HA-coated femoral stem
- 11 stem profile options
- 7-year clinical follow-up data
- Modular proximal-distal assembly
- Porous titanium augments
- Minimized fracture risk design
- Cemented and cementless options
Global Innovation Network
Our international network of R&D centers and academic partnerships drives continuous innovation in orthopedic implant technology.

3 R&D Centers Worldwide
Beijing • Shanghai • Sheffield, UK
International orthopedic research hub focusing on implant surface science, drug delivery, osteogenic factor integration, and novel bio-coating technologies.
Key Focus Areas
- China-UK co-development program
- European market access hub
- Multi-center clinical trials
Partnerships
Municipal government-recognized innovation platform for new orthopedic material development, porous titanium implant design, and manufacturing process optimization.
Key Focus Areas
- Material science research
- Manufacturing optimization
- Process innovation
Partnerships
Government-recognized platform advancing 3DACT™ platform development and translation of 3D printing innovations from laboratory to regulated clinical products.
Key Focus Areas
- 3D printing advancement
- Regulatory pathway support
- Clinical translation
Partnerships
AK Institute
Established 2012
Facilitate adoption of advanced orthopedic technologies through structured educational programs for surgeons, residents, and clinical staff.
Training Programs
- Cadaveric surgical skills workshops
- Robotic surgery simulation training
- ICOS personalized implant planning courses
- International exchange fellowships (UK facilities)
Listed on Hong Kong Stock Exchange
AK Medical Holdings Limited has been listed on the Main Board of the Hong Kong Stock Exchange since 2017, representing a milestone for China's orthopedic industry.
- Full Legal Name
- AK Medical Holdings Limited
- Stock Exchange
- Hong Kong Stock Exchange (HKEX) — Main Board
- Stock Code
- 01789.HK
- Year of Listing
- 2017
- Year of Founding
- 2003
- Headquarters
- Beijing, China
- Business Description
- Design, manufacture and sale of orthopedic implants, instruments, and digital surgery systems
Remuneration Committee
Established 2017. Primary mandate: recommend remuneration packages for executive directors and senior management.
Nomination Committee
Established 2017. Primary mandate: review Board composition and recommend candidates to fill vacancies.
Shareholder Communication
Corporate communications distributed in English and Chinese. Shareholders may elect hard copy or electronic receipt.
World Firsts in 3D-Printed Orthopedics
World's first clinically validated NMPA Class III registration for 3D-printed orthopedic implant
World's first Class III certificate for 3D-printed patient-matched orthopedic implants
World's first customized orthopedic implant product filing certificate
Get in Touch
Whether you're a healthcare institution, distributor, or surgeon interested in our products, we're here to help. Reach out to our global sales team.
No. 10 Baifuquan Road, Changping Science Park, Beijing 102200, China
+86 (010) 80109581
Sheffield, United Kingdom
Specializing in cementless biologic (HA-coated) hip implants and VPS coating systems for the European market.
5/F Manulife Place, 348 Kwun Tong Road, Kowloon, Hong Kong
For investor relations and shareholder communications regarding HKEX: 01789.HK.